Angers Ducs (FRA), SG Cortina (ITA), Gyergyoi HK (ROU), and HK Mogo (LAT) are set for the 2026 IIHF Continental Cup, Round 2, Group C.
The penultimate stage of the pan-European club competition will be held at Angers IceParc…

Angers Ducs (FRA), SG Cortina (ITA), Gyergyoi HK (ROU), and HK Mogo (LAT) are set for the 2026 IIHF Continental Cup, Round 2, Group C.
The penultimate stage of the pan-European club competition will be held at Angers IceParc…

AbbVie Announces Positive Topline Results from Phase 3 Pivotal Studies Evaluating Upadacitinib (RINVOQ®) in Adults and Adolescents with Vitiligo
NORTH CHICAGO, Ill., Oct. 29, 2025 /PRNewswire/ — AbbVie (NYSE: ABBV) today announced positive topline results from two replicate Phase 3 studies evaluating the safety and efficacy of upadacitinib (RINVOQ®;15 mg, once daily in adult and adolescent patients living with non-segmental vitiligo (NSV).1 NSV, the most common form of vitiligo (occurring in over 90% of patients), is characterized by symmetrical and bilateral white patches on both sides of the body.2-4
“Vitiligo is more than a skin condition – it’s a chronic autoimmune disease that can deeply affect a person’s confidence, identity and daily life,” said Kori Wallace, M.D., Ph.D., vice president, global head of immunology clinical development, AbbVie. “There are no approved systemic medical therapies for achieving re-pigmentation in vitiligo. These Phase 3 results represent a significant milestone in AbbVie’s commitment to supporting patients and expanding our immunology portfolio to deliver innovative solutions.”
The Total Vitiligo Area Scoring Index (T-VASI) is a tool that measures the extent of de-pigmentation over the entire body while F-VASI measures de-pigmentation of the face, an area among the most visible and psychosocially impactful for people living with NSV. Across both studies approximately 70% of subjects had T-VASI > 10 at baseline. In both studies, upadacitinib achieved the co-primary endpoints of T-VASI 50 (≥50% reduction from baseline in T-VASI score) and F-VASI 75 (≥75% reduction from baseline in F-VASI score) at week 48 versus placebo. Across both studies, statistically significant differences were observed with upadacitinib versus placebo in key ranked secondary endpoints, including F-VASI 50 at week 48.1
Key efficacy results are summarized below:
|
Phase 3 Efficacy Results1 |
||||
|
Study 1 |
Study 2 |
|||
|
Upadacitinib 15 mg (N=206), % |
Placebo (N=102), % |
Upadacitinib 15 mg (N=205), % |
Placebo (N=101), % |
|
|
Co-Primary Endpoints |
||||
|
T-VASI 50 at week 48 |
19.4 |
5.9 |
21.5 |
5.9 |
|
F-VASI 75 at week 48 |
25.2 |
5.9 |
23.4 |
6.9 |
|
Secondary Endpoints |
||||
|
F-VASI 50 at week 48 |
48.1 |
12.7 |
43.4 |
12.9 |
“For many people living with vitiligo, the journey is marked by uncertainty, frustration and a lack of medicines that can treat the disease systemically,” said Thierry Passeron, M.D., Ph.D., professor and chair, Department of Dermatology, Université Côte d’Azur. “These positive results indicate that targeting the underlying inflammation may offer a systemic treatment option which could help patients achieve visible results.”
The safety profile of upadacitinib in both studies was generally consistent with that observed in approved indications. No new safety signals were observed. Across both studies, the most frequent treatment-emergent adverse events (TEAEs) in the 48 weeks upadacitinib treatment groups were upper respiratory tract infection, acne and nasopharyngitis. In study 1, treatment-emergent serious adverse events (TESAEs) occurred in 3.9% and 4% of patients in the upadacitinib 15 mg and placebo groups, respectively. In study 2, TESAEs occurred in 2% and 1% of patients in the upadacitinib 15 mg and placebo groups, respectively. There were no adjudicated cases of any major cardiovascular event (MACE) or venous thromboembolism (VTE) in the studies. Three malignancy events were reported: one was reported in both placebo groups (1% each) of studies 1 and 2, and one was reported in the upadacitinib 15 mg group (0.5%) in study 1 (genital neoplasm). There were no deaths reported in the upadacitinib treated groups across both studies. There was one death reported in the placebo group in study 2.1
Use of upadacitinib in NSV is not approved and its safety and efficacy have not been evaluated by regulatory authorities.
About Vitiligo
Vitiligo is a chronic autoimmune disease characterized by the loss of pigment-producing cells (melanocytes), resulting in white patches of skin that can appear anywhere on the body and at any time. It is the most common depigmenting disorder worldwide, affecting approximately 0.5% to 2.3% of the global population. Non-segmental vitiligo (NSV) is the predominant form of the disease, accounting for roughly 84% of cases. NSV typically presents as symmetrical, bilateral patches on both sides of the body. While location varies, many patients report patches on critical areas such as the face, feet, hands and groin. The daily challenges of living with vitiligo can lead to mental health conditions, with a high prevalence of depression and anxiety.
About Viti-Up Clinical Trials
Upadacitinib M19-044 was conducted under a single protocol encompassing two replicate Phase 3 studies (Study 1 and Study 2) with independent randomization, investigative sites, data collection, analysis and reporting for each study. The trials were designed to evaluate the efficacy, safety and tolerability of upadacitinib in adult and adolescent patients (ages 12 and older) living with non-segmental vitiligo (NSV) who were eligible for systemic therapy. In Period A of both studies, participants were randomized in a 2:1 ratio to receive either upadacitinib 15 mg once daily or placebo for 48 weeks. Participants who completed Period A were eligible to enter Period B, a 112-week open-label extension in which all patients received upadacitinib 15 mg once daily. In total, Study 1 and Study 2 Periods A and B span 160 weeks. The two trials randomized 614 participants with NSV across 90 sites worldwide. More information on these trials can be found at www.clinicaltrials.gov (NCT06118411).
The co-primary endpoints were based on the achievement of Total Vitiligo Area Scoring Index (T-VASI) 50, defined as at least 50% reduction in T-VASI from baseline, at week 48, and the achievement of Facial Vitiligo Area Scoring Index (F-VASI) 75, defined as at least 75% reduction in F-VASI from baseline, at week 48 with the treatment of upadacitinib 15 mg compared with placebo in adults and adolescents with NSV.
The secondary endpoints include the achievement of F-VASI 50, defined as at least a 50% reduction in F-VASI from baseline, at week 48, and the achievement of F-VASI 75, defined as at least a 75% reduction in facial vitiligo area from baseline, at week 24. These endpoints were designed to assess the degree and timing of re-pigmentation on the face, an area among the most visible and psychosocially impactful for people living with NSV. Study 1 also included an analysis using 3D digital imaging to assess facial repigmentation, measured in a subset of the study population.
About RINVOQ® (upadacitinib)
Discovered and developed by AbbVie scientists, RINVOQ is a JAK inhibitor that is being studied in several immune-mediated inflammatory diseases. In human leukocyte cellular assays, RINVOQ inhibited cytokine-induced STAT phosphorylation mediated by JAK1 and JAK1/JAK3 more potently than JAK2/JAK2 mediated STAT phosphorylation. The relevance of inhibition of specific JAK enzymes to therapeutic effectiveness and safety is not currently known.
Upadacitinib (RINVOQ) is being studied in Phase 3 clinical trials for alopecia areata, hidradenitis suppurativa, Takayasu arteritis, systemic lupus erythematosus and vitiligo.
RINVOQ (upadacitinib) U.S. Uses and Important Safety Information5
RINVOQ is a prescription medicine used to treat:
It is not known if RINVOQ is safe and effective in children with ankylosing spondylitis, non-radiographic axial spondyloarthritis, ulcerative colitis, or Crohn’s disease.
It is not known if RINVOQ is safe and effective in children under 12 years of age with atopic dermatitis.
It is not known if RINVOQ LQ is safe and effective in children with atopic dermatitis.
RINVOQ/RINVOQ LQ is a prescription medicine used to treat:
It is not known if RINVOQ/RINVOQ LQ is safe and effective in children under 2 years of age with polyarticular juvenile idiopathic arthritis or psoriatic arthritis.
IMPORTANT SAFETY INFORMATION FOR RINVOQ/RINVOQ LQ (upadacitinib)
What is the most important information I should know about RINVOQ*?
RINVOQ may cause serious side effects, including:
Do not take RINVOQ if you are allergic to upadacitinib or any of the ingredients in RINVOQ. See the Medication Guide or Consumer Brief Summary for a complete list of ingredients.
What should I tell my HCP BEFORE starting RINVOQ?
Tell your HCP if you:
|
̶ Fever, sweating, or chills ̶ Shortness of breath ̶ Warm, red, or painful skin or sores on your body |
̶ Muscle aches ̶ Feeling tired ̶ Blood in phlegm ̶ Diarrhea or stomach pain |
̶ Cough ̶ Weight loss ̶ Burning when urinating or urinating more often than normal |
Tell your HCP about all the medicines you take, including prescription and over-the-counter medicines, vitamins, and herbal supplements. RINVOQ and other medicines may affect each other, causing side effects.
Especially tell your HCP if you take:
If you are not sure if you are taking any of these medicines, ask your HCP or pharmacist.
What should I avoid while taking RINVOQ?
Avoid food or drink containing grapefruit during treatment with RINVOQ as it may increase the risk of side effects.
What should I do or tell my HCP AFTER starting RINVOQ?
|
̶ Swelling ̶ Pain or tenderness in one or both legs |
̶ Sudden unexplained chest or upper back pain ̶ Shortness of breath or difficulty breathing |
What are other possible side effects of RINVOQ?
Common side effects include upper respiratory tract infections (common cold, sinus infections), shingles (herpes zoster), herpes simplex virus infections (including cold sores), bronchitis, nausea, cough, fever, acne, headache, swelling of the feet and hands (peripheral edema), increased blood levels of creatine phosphokinase, allergic reactions, inflammation of hair follicles, stomach-area (abdominal) pain, increased weight, flu, tiredness, lower number of certain types of white blood cells (neutropenia, lymphopenia, leukopenia), muscle pain, flu-like illness, rash, increased blood cholesterol levels, increased liver enzyme levels, pneumonia, low number of red blood cells (anemia), and infection of the stomach and intestine (gastroenteritis).
A separation or tear to the lining of the back part of the eye (retinal detachment) has happened in people with atopic dermatitis treated with RINVOQ. Call your HCP right away if you have any sudden changes in your vision during treatment with RINVOQ.
Some people taking RINVOQ may see medicine residue (a whole tablet or tablet pieces) in their stool. If this happens, call your HCP.
These are not all the possible side effects of RINVOQ.
How should I take RINVOQ/RINVOQ LQ?
RINVOQ is taken once a day with or without food. Do not split, crush, or chew the tablet. Take RINVOQ exactly as your HCP tells you to use it. RINVOQ is available in 15 mg, 30 mg, and 45 mg extended-release tablets. RINVOQ LQ is taken twice a day with or without food. RINVOQ LQ is available in a 1 mg/mL oral solution. RINVOQ LQ is not the same as RINVOQ tablets. Do not switch between RINVOQ LQ and RINVOQ tablets unless the change has been made by your HCP.
*Unless otherwise stated, “RINVOQ” in the IMPORTANT SAFETY INFORMATION refers to RINVOQ and RINVOQ LQ.
This is the most important information to know about RINVOQ. For more information, talk to your HCP.
You are encouraged to report negative side effects of prescription drugs to the FDA. Visit www.fda.gov/medwatch or call 1-800-FDA-1088.
If you are having difficulty paying for your medicine, AbbVie may be able to help. Visit AbbVie.com/PatientAccessSupport to learn more.
Please click here for the Full Prescribing Information and Medication Guide.
Globally, prescribing information varies; refer to the individual country product label for complete information.
About AbbVie
AbbVie’s mission is to discover and deliver innovative medicines and solutions that solve serious health issues today and address the medical challenges of tomorrow. We strive to have a remarkable impact on people’s lives across several key therapeutic areas including immunology, oncology, neuroscience and eye care – and products and services in our Allergan Aesthetics portfolio. For more information about AbbVie, please visit us at www.abbvie.com. Follow @abbvie on LinkedIn, Facebook, Instagram, X (formerly Twitter) and YouTube.
Forward-Looking Statements
Some statements in this news release are, or may be considered, forward-looking statements for purposes of the Private Securities Litigation Reform Act of 1995. The words “believe,” “expect,” “anticipate,” “project” and similar expressions and uses of future or conditional verbs, generally identify forward-looking statements. AbbVie cautions that these forward-looking statements are subject to risks and uncertainties that may cause actual results to differ materially from those expressed or implied in the forward-looking statements. Such risks and uncertainties include, but are not limited to, challenges to intellectual property, competition from other products, difficulties inherent in the research and development process, adverse litigation or government action, changes to laws and regulations applicable to our industry, the impact of global macroeconomic factors, such as economic downturns or uncertainty, international conflict, trade disputes and tariffs, and other uncertainties and risks associated with global business operations. Additional information about the economic, competitive, governmental, technological and other factors that may affect AbbVie’s operations is set forth in Item 1A, “Risk Factors,” of AbbVie’s 2024 Annual Report on Form 10-K, which has been filed with the Securities and Exchange Commission, as updated by its Quarterly Reports on Form 10-Q and in other documents that AbbVie subsequently files with the Securities and Exchange Commission that update, supplement or supersede such information. AbbVie undertakes no obligation, and specifically declines, to release publicly any revisions to forward-looking statements as a result of subsequent events or developments, except as required by law.
References:
Contacts:
SOURCE AbbVie
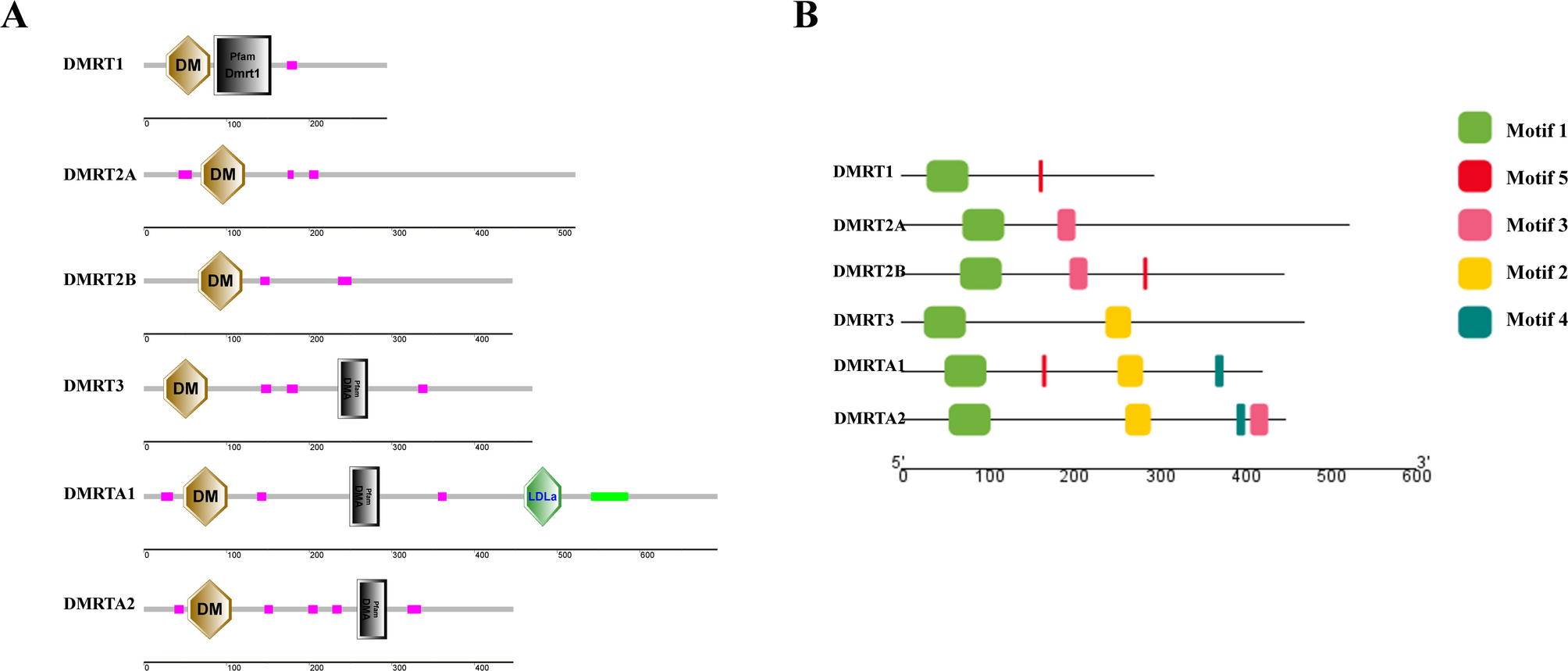
Kikuchi K, Hamaguchi S. Novel sex-determining genes in fish and sex chromosome evolution. Dev Dyn. 2013;242(4):339–53.
Google Scholar
Kang Y, Guan GJ, Hong YH. Insights of sex…
With populations aging rapidly worldwide, heart failure has become one of the most pressing medical and social challenges. Older patients face not only a high mortality risk but also losses in muscle strength, mobility, and…

On the topic of debutants, Emily Harrop will hope to write history as ski mountaineering is held at the Olympic Winter Games for the first time. Born in the French Alps to British parents, can she achieve a golden debut on the snow?
The series…

Proteins play a key role not only in the body, but also in medicine: they either serve as active ingredients, such as enzymes or antibodies, or they are target structures for drugs. The first step in developing new therapies is…

Developed in collaboration with Janes, the IBM Defense Model delivers actionable, defense-specific insights in secured environments
Oct 29, 2025
WASHINGTON, Oct. 29, 2025 /PRNewswire/ — IBM (NYSE: IBM) today announced the general availability of the IBM Defense Model, a purpose-built AI model designed to deliver reliable intelligence for defense and national security. Developed together with Janes, a leading provider of open-source defense intelligence, the IBM Defense Model combines IBM’s enterprise-grade AI technology with Janes domain-specific data to empower agencies to make decisions with speed, precision and confidence in secured, mission-critical environments.

Unlike general-purpose large language models, the IBM Defense Model is optimized for defense-specific tasks and deployable in air-gapped, classified, and edge settings. The model aligns with IBM’s focus on smaller fit-for-purpose, open-source AI models and datasets – developed and fine-tuned for specific domains and use cases – that can deliver exceptional value and drive innovation. Built on IBM’s Granite foundation models and delivered via IBM watsonx.ai, the solution supports planning, reporting, and strategy.
“Defense organizations need AI they can trust – solutions that deliver accurate insights without compromising security or ethics,” said Vanessa Hunt, General Manager, Technology, U.S. Federal Market for IBM. “The IBM Defense Model provides a fit-for-purpose capability that accelerates mission planning and enhances operational readiness, while reinforcing IBM’s commitment to responsible AI.”
Key features and benefits:
“Our collaboration with IBM brings together Janes trusted defense intelligence and IBM’s advanced AI capabilities,” said Blake Bartlett, CEO for Janes. “This model helps to ensure defense organizations can access timely, relevant insights in secured environments, helping them make informed decisions with confidence.”
The IBM Defense Model is now available. For more information, visit https://www.ibm.com/products/watsonx-ai/defense-model.
About IBM
IBM is a leading provider of global hybrid cloud and AI, and consulting expertise. We help clients in more than 175 countries capitalize on insights from their data, streamline business processes, reduce costs and gain the competitive edge in their industries. Thousands of governments and corporate entities in critical infrastructure areas such as financial services, telecommunications and healthcare rely on IBM’s hybrid cloud platform and Red Hat OpenShift to affect their digital transformations quickly, efficiently and securely. IBM’s breakthrough innovations in AI, quantum computing, industry-specific cloud solutions and consulting deliver open and flexible options to our clients. All of this is backed by IBM’s long-standing commitment to trust, transparency, responsibility, inclusivity and service. Visit www.ibm.com
Media contact
Banks Willis
banks.willis@ibm.com
SOURCE IBM

Former MP Mhairi Black joins the cast of new BBC high-stakes legal drama Counsels playing a police detective in her first acting role for television. Mhairi will star in four episodes of the eight-part series and starts filming next week for the…

The sea, in the plays of Eugene O’Neill, is never neutral: It is fate, peril, self-delusion, rebirth, truth. How fitting, then, that it is on a New York waterfront that I meet Michelle Williams, the star of a new production of Anna Christie…