Harmonized Landsat and Sentinel-2 PI Christopher Neigh shares milestones and a vision for the future
In 2025, the Harmonized Landsat and Sentinel-2 (HLS) program established itself as a cornerstone for global medium-resolution optical Earth…
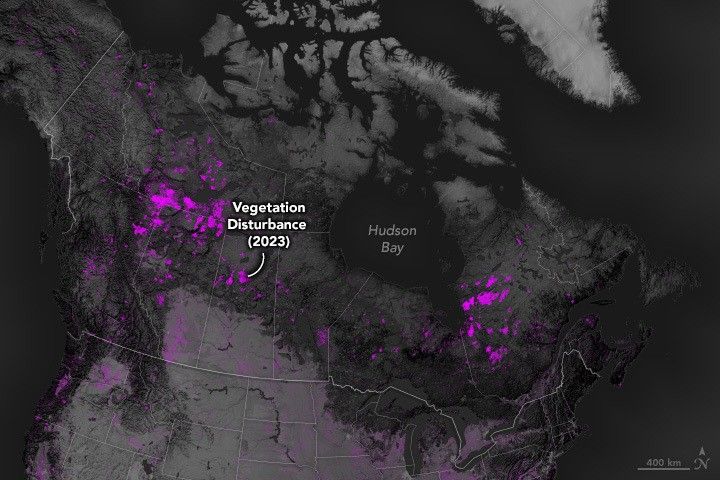
In 2025, the Harmonized Landsat and Sentinel-2 (HLS) program established itself as a cornerstone for global medium-resolution optical Earth…