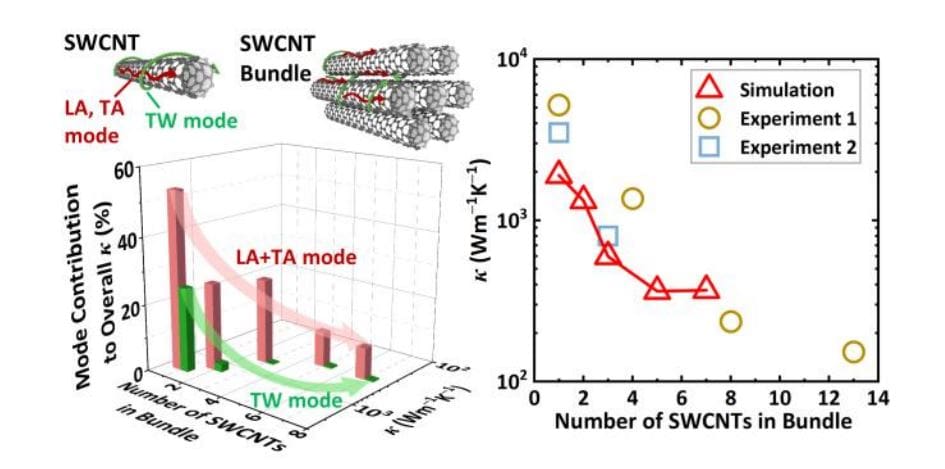
The dramatic reduction in thermal conductivity within bundles of single-walled carbon nanotubes (SWCNTs) has long puzzled scientists, hindering their application in advanced thermal management systems. Now, Feng Tao, Xiaoliang Zhang, Dawei…
Predicting SWCNT Bundle Thermal Conductivity Enables New Materials Design With Machine Learning