Primary findings
This study aimed to investigate UOD-induced thermomechanical effects on bone microstructure and brain tissue preservation in neuro-spine surgery analogs. We found that the bone healing is comparable between UOD and HSB defects, as shown by micro-CT images. However, histological analyses revealed laminated-layered structural changes to the bone tissues adjacent to the UOD cutting defects. Moreover, abrupt temperature spikes of UOD use were noticed during the UOD cutting, and we have demonstrated neurothermal sequelae of UOD osteotomy that have not been reported elsewhere. Specifically, the mice cut by UOD showed mildly altered parietal lobe functions, and histological analyses showed brain defects sustained for more than 8 weeks, revealing a previously uncharacterized risk profile.
Thermal dynamics in ultrasonic osteotome device applications
Our study demonstrated transient localized temperature spikes (> 60 °C) during UOD osteotomy using thermal imaging despite irrigation (Supplementary Material 1). This observation aligns with prior findings reporting elevated temperature during UOD procedures23,24,25,26.
For instance, the observed temperature spikes align with Suzuki et al.’s findings in porcine bone, where UODs exceeded HSB temperatures by 28 °C27. Regarding in vivo experiments, transient tip temperatures exceeding 85 °C were recorded when UODs were cutting the inner cortex of the lamina during laminectomy, and after laminectomy, the average temperature of the inner cortex of the lamina was increased up to 11.4°C28. Without irrigation, the temperature of the cutting surface can reach up to 150 °C29.
Increased temperature, known as heat shock or thermal insults during the bone osteotomy may hence result in thermal necrosis30causing an immediate necrotic response in bone cells or can induce longer apoptotic responses, with thresholds of 47 °C for 60 s or 50 °C for 30 s31,32,33. Variations in the temperature thresholds may be partly attributable to bone quality, as cancellous bone suffered greater damage from thermal treatment than cortical bone34.
Cellular responses are triggered depending on the extent and duration of exposure to heat shock, leading to cell death by either necrosis or apoptosis, as shown in Figure S4. Thermal osteonecrosis consists of bone cell death and subversion of endosteal architecture32. Consequently, the bone will be resorbed and replaced with fibrous connective tissue33.
Contemporary irrigation systems have markedly reduced the cutting temperature by using sharper cutting tips, constant irrigation, and controlled drilling speeds, among others35. Nevertheless, our study demonstrates that novel cooling methods still need further colling the cutting temperature to avoid osteonecrosis.
Novel findings in bone structural alterations after piezosurgery
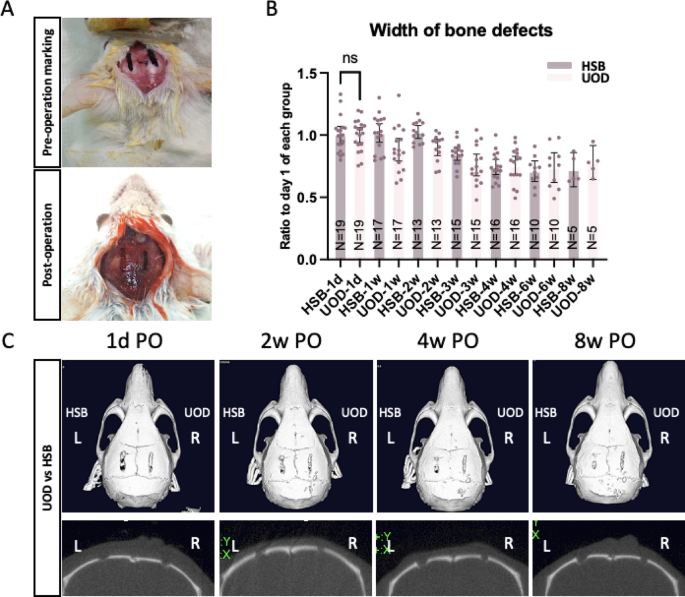
On the one hand, our results are consistent with previous findings in several aspects36. First, the use of UOD can achieve a smooth cutting surface, and the bone defects can heal properly after the cutting, no worse than the HSB (Fig. 1B-C)36. Nevertheless, on the other hand, different from other studies, our H&E staining results show that the bones adjacent to the UOD cuts showed laminated-layered structural changes (Fig. 2), which is also true when cut with other UODs (Figure S2).
Previous studies have presented mixed conclusions regarding the bone-cutting performance and bone-healing effects of UODs compared to conventional HSBs. For example, UODs were reported to have a superior bone healing profiles36,37,38. Specifically, UODs could produce a smooth cutting surface devoid of micro-fractures36 compared to HSB, facilitate bone healing compared to trephine burs37and favor the preservation of bone regions adjacent to osteotomies, although UODs may trigger variations in gene expression38.
Nevertheless, other studies have stressed that replacing HSBs with UODs could not improve bone healing8,9,39,40and that using UODs would lead to distinct bone healing patterns. For instance, UODs could still induce micro-cracks formation, and different cutting modes or bone structures may yield irregular cutting performance39. Moreover, Bergonse Pereira Pedriali et al. reported that UODs delayed initial bone ossification, though fewer postoperative inflammatory responses were elicited40. No significant differences between rotating drills/saws and piezosurgery were reported by Esteves et al.8 and Ma et al.9and increased bone remodeling activity was found in the piezoelectric surgery group9.
Novel findings in CNS structural and functional alterations after receiving piezosurgery
While prior research suggested UODs have limited nerve damage potential and eventual recovery in peripheral nerves, our findings reveal significant and lasting structural and functional alterations in the central nervous system (CNS) following calvarial bone cutting.
Critically, we observed pathological structural changes in the brain tissues underlying the bone cut, including edema, structural defects, and hyalinization (Fig. 2), indicative of thermal coagulative necrosis and cell death41. The histomorphometric findings align with hyperthermia-induced brain tissue damages, evidenced by apoptotic cells, fragmented nuclei, and hyalinized zones likely to be aggregated proteins from heat-denatured cytoskeletal components and hydrophobic interactions between damaged proteins42,43.
These irreversible alterations correlate with functional deficits, demonstrating that the soft tissue selectivity of UODs is limited, and the damage to soft tissues may still occur during the cutting; this denotes a novel in vivo observation contrasting with ex vivo or peripheral nerve studies.
The parietal cortex of rodents integrates multisensory information and coordinates spatial navigation, which can be investigated by tests assessing the Somatosensory Processing (viz., Adhesive Removal Test, Von Frey Filaments) and Spatial Cognition and Motor Integration (viz., Whisker-Nuisance Test, Puzzle Box Test)44,45,46,47,48. Nevertheless, the cylinder test was employed in this study as it can assess the distinction of the bilateral somatosensory processing and spatial cognition of parietal lobes20,21which are manifested as the contralateral motor functions.
For CNS, past animal safety studies (Supplementary Table 1) using the spinal cord of cats49,50 and dogs51 using extreme exposure parameters (viz., prolonged direct contact, high energy levels not clinically used) have shown that there is an energy- and duration-dependent damage to the soft tissues with the use of UODs5. Although these studies stressed that the required injury parameters exceed clinical practice and suggested peripheral nerve injury was often transient with anticipated recovery5,51our observation of hyalinization and associated brain necrosis provides direct histopathological evidence that clinically encountered thermal energy transfer (as seen in device tip spikes > 60–70 °C) can inflict irreversible damage to adjacent vulnerable CNS tissues, underscoring a critical safety concern.
For peripheral nervous tissues, prior investigations6 have found that direct exposure of a peripheral nerve to Piezosurgery, even in the worst-case scenarios, did not dissect the nerve transection but did induce some structural and functional damage in rat models, where the perineurium was damaged, and the functional test of the rats interfered6,7.
Interestingly, after a thorough comparison with findings by Schaeren et al., we noticed that the UODs exerted similar structural alterations to the nerves, characterized by a multilayered wave-like morphology in their study6which is consistent with the multilayered wave-like morphology observed in the H&E brain tissues in this study.
Clinical interpretations and future directions
Bone healing after UOD osteotomy is influenced by device features and patient-specific factors, such as bone type (trabecular vs. cortical) and density (whether osteoporotic), as illustrated in Fig. 42,10,11,12,14. Suboptimal thermal management during osteotomy can impede healing, and future research should explore strategies to reduce heat production, optimize cutting parameters, and enhance bone healing, particularly in patients with osteoporosis or systemic comorbidities.
The factors influencing bone healing when using ultrasonic osteotomy devices.
Removal of bony elements in regions with complex anatomical structures is common in spine and cranial surgery; for instance, laminectomy and decompression of multilevel compressive myelopathy are technically challenging using traditional instruments in a highly narrow epidural space.
Although systematic reviews have affirmed that laminectomy done with UODs was found to be safe, rapid, and effective with low blood loss52and spinal surgeries with a UOD were reported to show a reduction in osseous bleeding53durotomy and soft tissue damage still occur from time to time16.
As shown in ex vivo cutting experiments, UOD generates significant localized heat at the cutting site, and our study has demonstrated that UOD cutting could cause underlying brain tissue damage in the mouse model. The heat generated from UOD cutting may be transferred to the brain tissues, causing thermal osteonecrosis, which may be attributable to the limited insulation that the thin mouse calvarial bone offered. Therefore, this finding may provide important clinical messages that extreme caution should be exercised when applying the UODs to remove bone where distance is limited and delicate tissues are in proximity, such as pediatric patients, skull base procedures, or surgeries near neurovascular structures, especially when these structures are known to be compromised or damaged.
Strength and limitation
This study is the first to directly compare the bone healing effects and brain tissue damage of UODs with HSB regarding brain tissue damage and behavioral tests using split calvarial bone defect models. However, limitations exist.
First, the assessment of bone healing and soft tissue damage was limited to macroscopic and microscopic phenotypic levels without delving into molecular changes. Moreover, the density calibration was not conducted upon micro-CT scanning. Hence, density-based quantification was not possible, and future studies are needed to further validate the density-related damage and findings. Second, the use of mice, whose bone density and structure differ significantly from humans, limits the generalizability of our findings. However, the murine calvarial defect model remains valuable for simulating delicate bone-cutting procedures adjacent to central nervous tissue, such as in spine surgeries where durotomy may occur.
Volumetric bone regeneration parameters such as bone volume fraction (BV/TV) or trabecular morphology were not investigated in this study, and the lack of microarchitectural analysis may impede the comprehensive characterization of mineralization patterns and bone quality maturation following thermal exposure. Future studies incorporating micro-CT analysis could provide critical insights into whether transient thermal spikes during UOD osteotomy would induce lasting alterations in cortical porosity or trabecular connectivity.
Third, this study only conducted H&E staining, which may not distinctly distinguish the newly generated bone structure as other histomorphometric approaches54,55,56and only the coronal planes of the calvarial bone sections were conducted; hence, the measurements may be biased. Additionally, morphometric measurements were potentially biased because only coronal sections were analyzed, and the irregular shapes of the brain defects, combined with tissue distortion during compression and mounting, complicated accurate quantification. To minimize these biases, defect areas were determined by averaging three measurements. The operation duration was measured via an operation video clip from cutting to the end of the osteotomy, excluding skin excision and suture time. As the experiment subjects and osteotomy size are small, the operation duration measurements may not reflect the true surgical situation.
Moreover, although device parameters were standardized at single output settings in this study throughout the experiments, with Sonicmed at 60% maximal cutting power and Misonix and Sonopet using clinically recommended presets, this approach does not account for performance variability across different power levels or irrigation regimens. Still, different setting parameters of each device were not tested, which may lead to difficulty in comparison and potential biases. Hence interpretation should be cautious and future studies are urged.
Additionally, the sole reliance on thermal cameras to measure and record procedural temperature changes may not fully capture the heat transduction dynamics between the cutting tip and the target tissues during UOD procedures. Consequently, the potential effects of abrupt temperature changes during UOD cutting on bone healing and soft tissues warrant further investigation. These methodological constraints necessitate cautious interpretation of our results and validation studies in models more closely approximating human skeletal properties.