Home
»
TRAVEL ALERT
»
NEW CDC GUIDELINES: Guyana Joins Bangladesh, Cuba, Maldives, Mali, Mauritania, Samoa, Sudan, Timor-Leste,Vietnam And More Nations in Dengue Fever Alert, Raising Urgent Travel Concerns Over Rising Transmission Rates
…

Terahertz (THz) radiation is increasingly explored for biomedical applications, however, its non-thermal effects on cellular metabolism and regulatory networks remain insufficiently characterized. This study aimed to investigate how 2.3…
In 2024, the New England Journal of Medicine published results from the groundbreaking FLOW study, showing that the glucagon-like peptide-1 (GLP-1) receptor agonist semaglutide reduces the risk of clinically important kidney outcomes and death…
Elendu, C. et al. Global perspectives on the burden of sexually transmitted diseases: A narrative review. Medicine 103 (20), e38199 (2024).
World Health Organization (WHO). Sexually transmitted…

SUDAN is in the midst of an outbreak of acute hepatitis among displaced populations, highlighting the vulnerability of communities in conflict-affected regions. Acute hepatitis, an inflammatory liver condition that can progress to liver…

27 Mars 2026
27The results of the recent IARC Monographs evaluation of three chemicals – tris(chloropropyl) phosphate (TCPP), butyraldehyde, and cumyl hydroperoxide – have now been published in The…
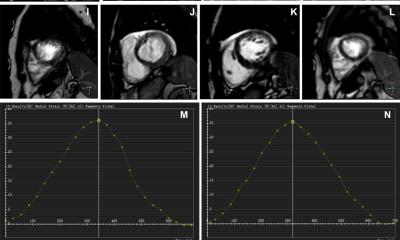
The AI-CS framework offers a promising alternative for cardiac MRI examinations in the clinical setting, where long acquisition time remains a major challenge
Nan Zhang
“For patients with severe arrhythmias, breath-holding can be particularly…

Researchers from the Federal University of São Carlos (UFSCar) in Brazil, working with University College London (UCL) in the United Kingdom, found that having both excess abdominal fat and reduced muscle mass significantly raises the risk of…